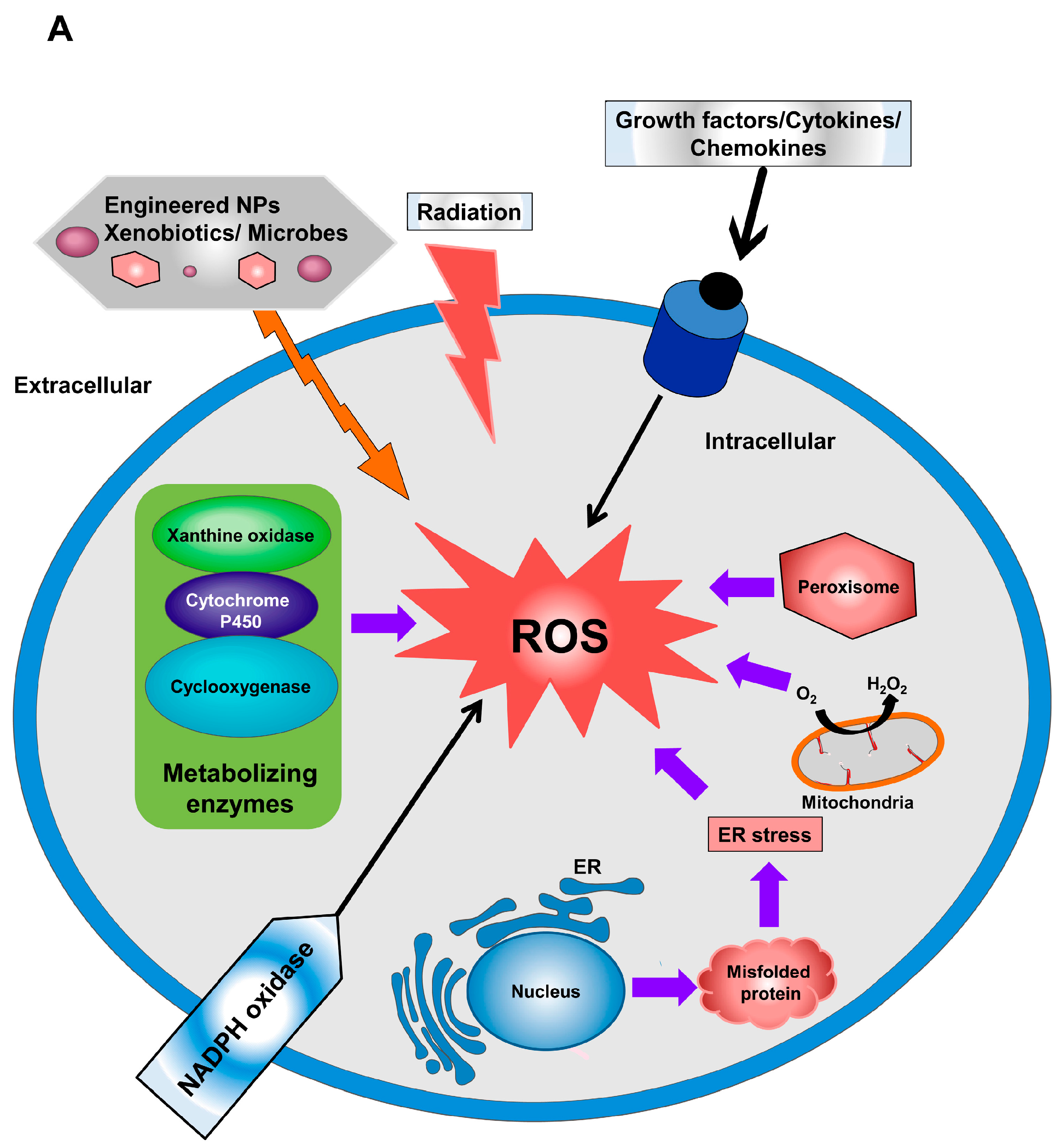
This balance may become disturbed under conditions of hyperoxia, inflammation, or ischemia-reperfusion (excessive generation of ROS) or in the presence of limited or impaired antioxidant defenses. Failure to adequately sequester or store iron could be a developmental liability in premature infants with relative deficiencies in iron carriers, such as transferrin ( 11).Ī delicate balance exists between ROS production and the antioxidant defenses that protect cells in vivo. In contrast, indirect evidence has linked the presence of free iron with increased protein carbonyl formation in patients treated with high concentrations of supplemental oxygen. Although free iron (unbound to ferritin or heme, for example) has been documented in the circulating plasma of preterm newborns, the detrimental effects of free iron or other metals have not been definitively established in newborns ( 9, 10). The burden of ROS can be further amplified by the presence of “free” metals, such as iron, copper, and manganese, which can be released from metalloprotein complexes. ROS are also important in the regulation of nitric oxide bioavailability, dramatically influencing airway and vascular reactivity ( 2). − also arises from NADPH oxidases ( NOX 1–3 typically at low levels in smooth muscle and vascular endothelium), dual oxidases 1 and 2, and NOX 4 (epithelial cells) ( 8). 
In addition to ROS generated as a “byproduct” of cellular respiration, endogenous production of O 2
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
